A testament to discovery
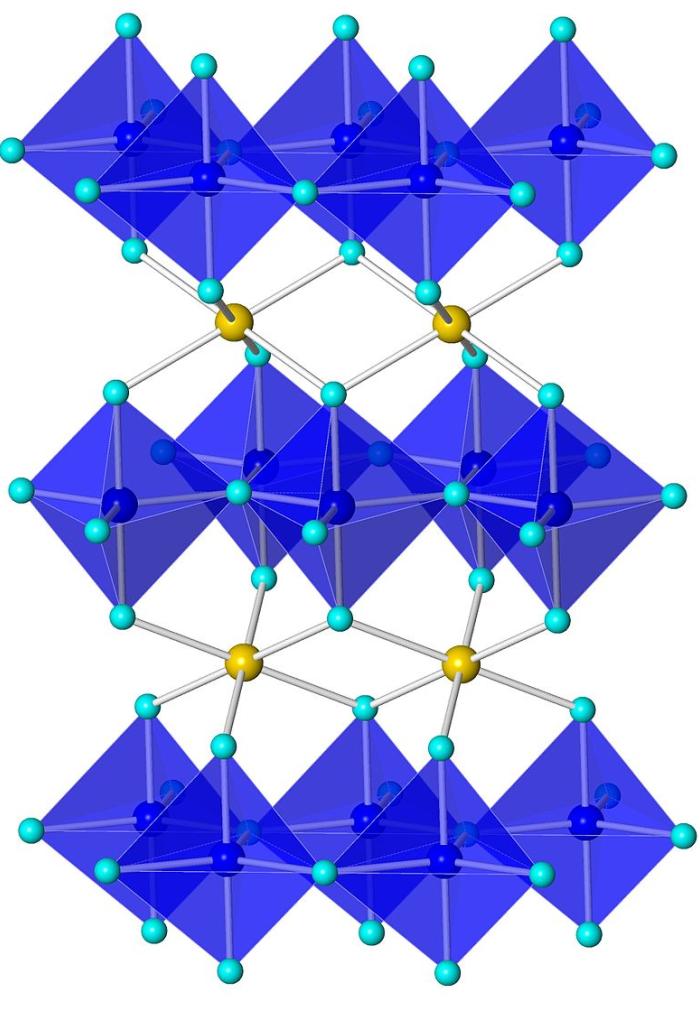
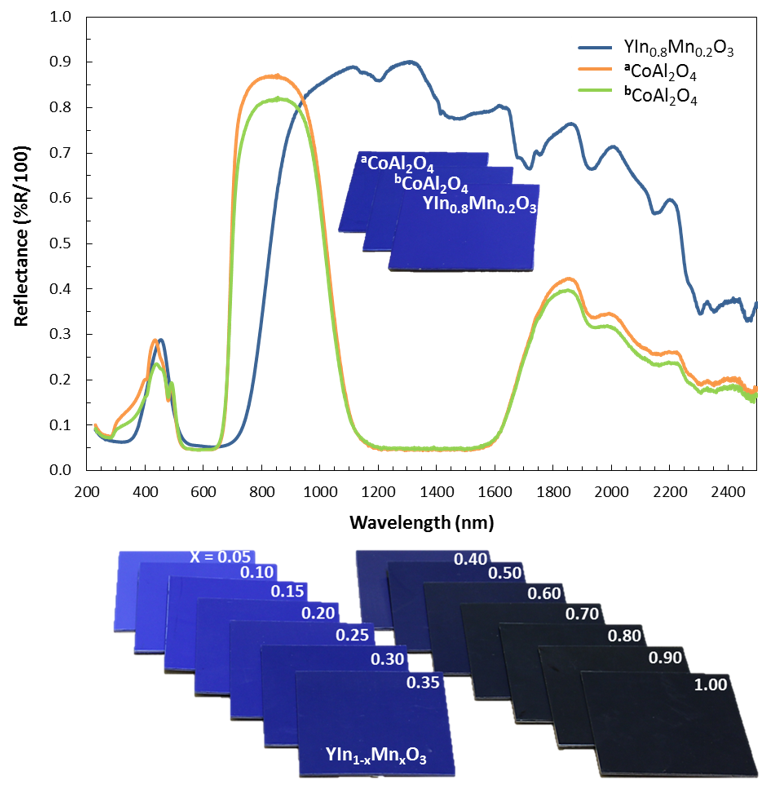
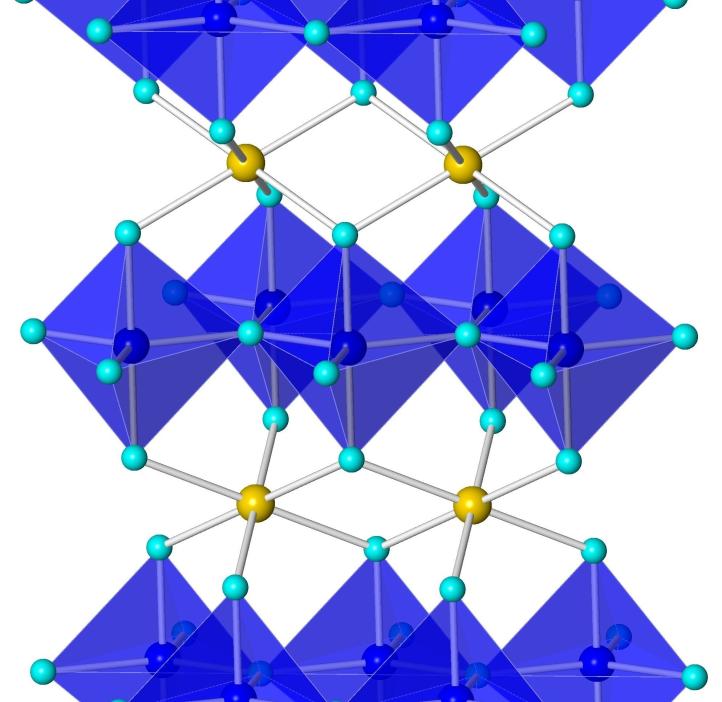
YInMn Blue, or "MasBlue" as it is commonly referred to at Oregon State University, is a serendipitous discovery of a bright blue pigment by scientists led by Mas Subramanian at OSU while researching materials for electronics applications. The pigment contains the elements Yttrium, Indium, Manganese, and Oxygen.